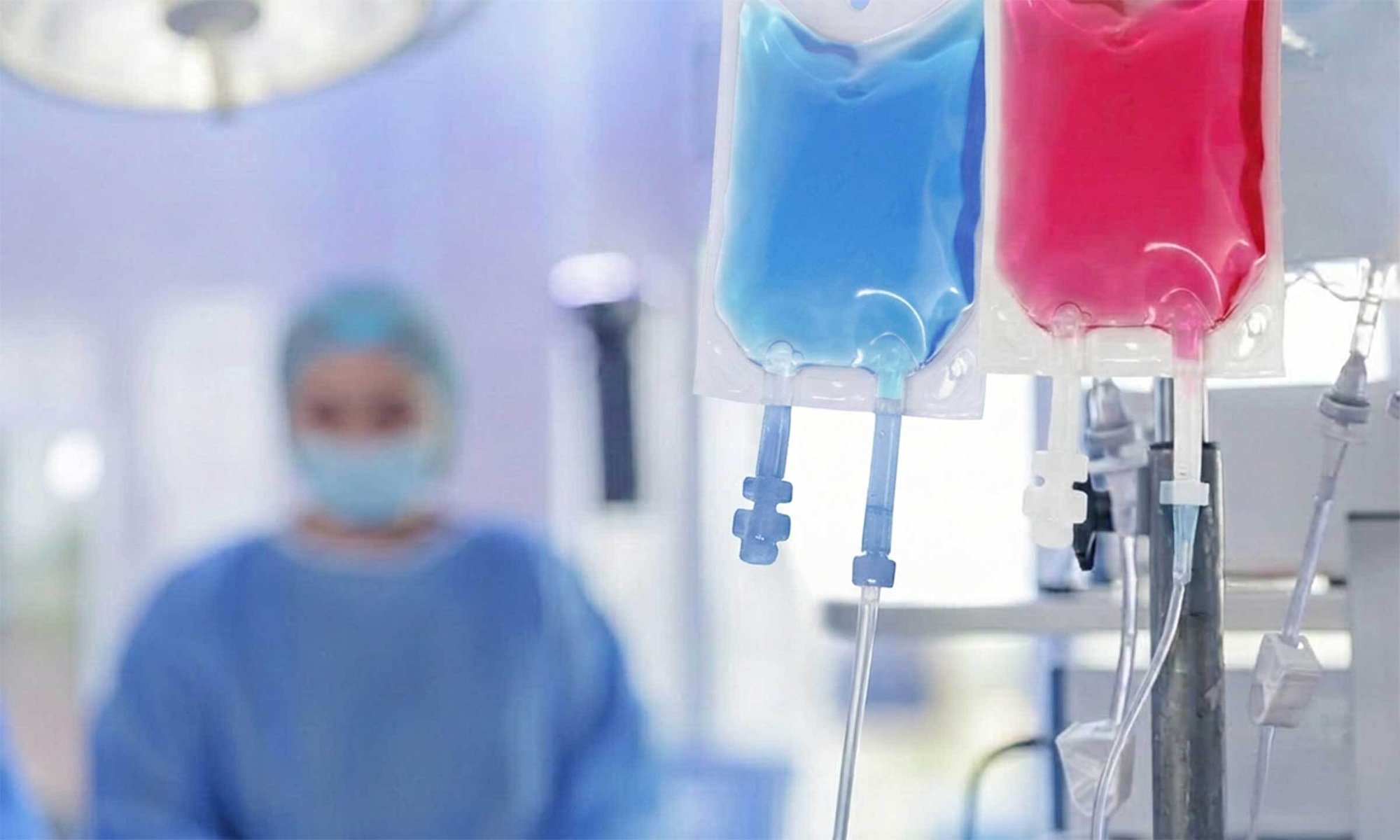
Non-PVC multi-layer IV Bags
Why choose GVS Non-PVC Multi-layer IV Bags
- Reduced drug loss for sensitive and lipophilic medications
- Lower DEHP exposure for high‑risk patient populations
- More predictable infusion performance
- Regulatory‑aligned material selection
- Improved environmental profile
For decades, PVC has been widely used in IV bags. However, its reliance on plasticizers such as DEHP — a substance not chemically bound to the polymer — has raised growing concerns around patient exposure, drug stability, and regulatory risk.
Non‑PVC Multi‑layer IV Bags are now setting the reference standard for safer, more consistent infusion therapy.
DEHP can migrate from PVC materials into infusion solutions, especially during long‑duration therapies or when lipid‑based drugs are used. Clinical and regulatory bodies have identified specific risk for vulnerable patient groups, including neonates, pediatric patients, pregnant women, and oncology patients receiving repeated infusions.
Non-PVC Multi-layer IV bags eliminate this exposure pathway, supporting safer care environments without compromising performance.
Supporting safer and more responsible material choices
PVC presents challenges at end‑of‑life due to chlorine chemistry and additive complexity. In contrast, non‑PVC materials such as polyolefins:
- Contain no chlorine
- Offer improved recyclability potential
- Reduce the risk of hazardous emissions
Non-PVC Multi-layer IV bags align with sustainability roadmaps and ESG‑driven procurement strategies increasingly adopted by healthcare organizations.


More reliable drug delivery, less variability
Certain drugs, particularly lipophilic or long‑infusion medications — are known to interact with PVC materials. This interaction can lead to drug sorption, reducing the delivered dose and introducing variability into therapy.
Non‑PVC multi‑layer bag structures significantly reduce this effect, helping to:
- Maintain intended drug concentration
- Improve dose accuracy
- Reduce the need for overfilling or compensatory dosing
The result is more predictable therapy, especially in critical and continuous infusion settings.
Consistent behavior throughout storage and use
Non-PVC Multi-layer IV Bags materials offer more stable mechanical and chemical behavior over time. By eliminating plasticizer migration and reducing material aging effects, these bags support:
- Consistent transparency and handling
- Reliable performance after sterilization
- Greater confidence during pharmacy preparation and clinical use
Designed for evolving regulatory expectations
Regulatory scrutiny of DEHP and plasticized PVC continues to increase worldwide. Non-PVC Multi-layer IV bags support alignment with:
- EU REACH requirements and long‑term phase‑out timelines
- FDA recommendations to limit phthalate use
- Extractables & leachables risk‑management strategies
Choosing Non-PVC Multi-layer IV Bags materials helps future‑proof infusion systems against tightening compliance requirements.
From PVC to Non‑PVC Multi-layer IV Bags
End‑to‑end expertise for future‑ready infusion solutions
GVS supports pharma companies and CMOs in the transition to Non-PVC Multi-layer IV bags with deep expertise in filtration, medical materials, and integrated manufacturing.
Why GVS
- Proven IV filtration and medical‑grade material know‑how
- PVC‑free bag platforms for LVPs, chemo, TPN, and critical therapies
- Fully integrated manufacturing, from films to components
- Designed to meet evolving DEHP, E&L, EU REACH, and USP expectations